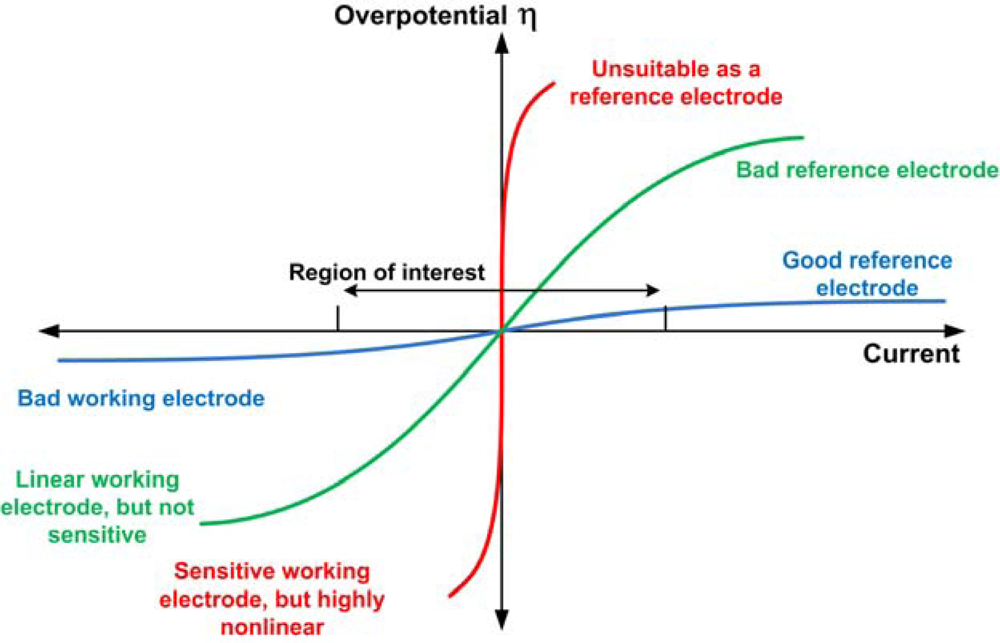
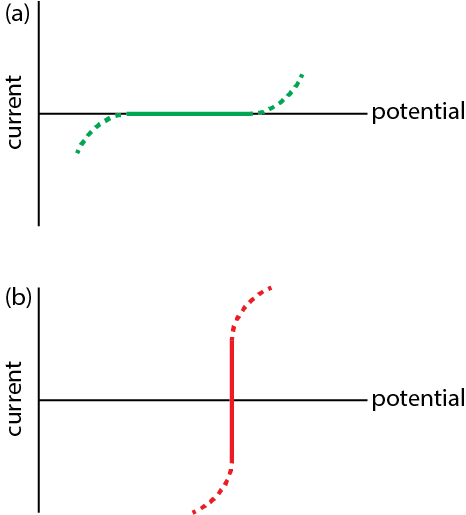
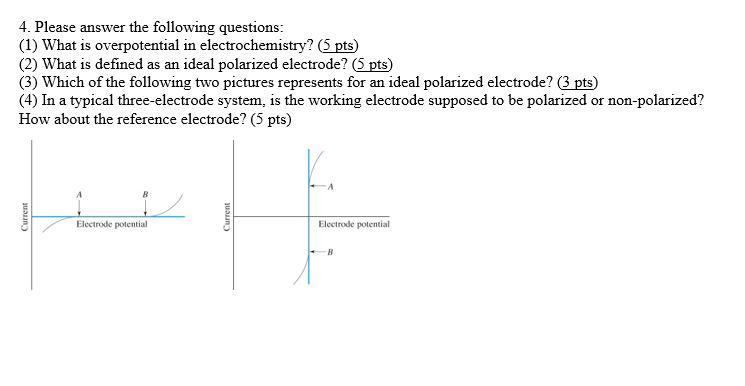
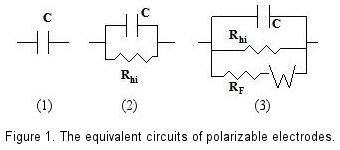
The role of reversible and polarizable surface charge in the electro-sorption of NaCl electrolyte onto activated carbon-graphite electrode - ScienceDirect

Understanding the Electric Double-Layer Structure, Capacitance, and Charging Dynamics | Chemical Reviews

Difference Between Polarizable and Non Polarizable Electrode | Compare the Difference Between Similar Terms
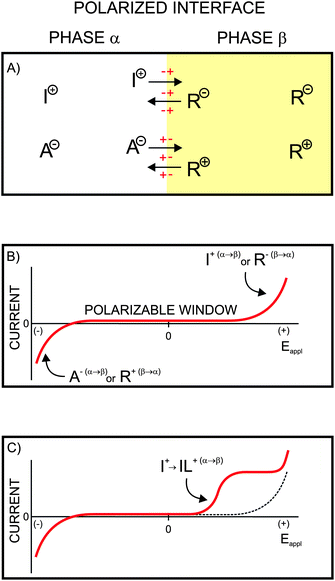
Dynamic electrochemistry with ionophore based ion-selective membranes - RSC Advances (RSC Publishing) DOI:10.1039/C3RA43751E
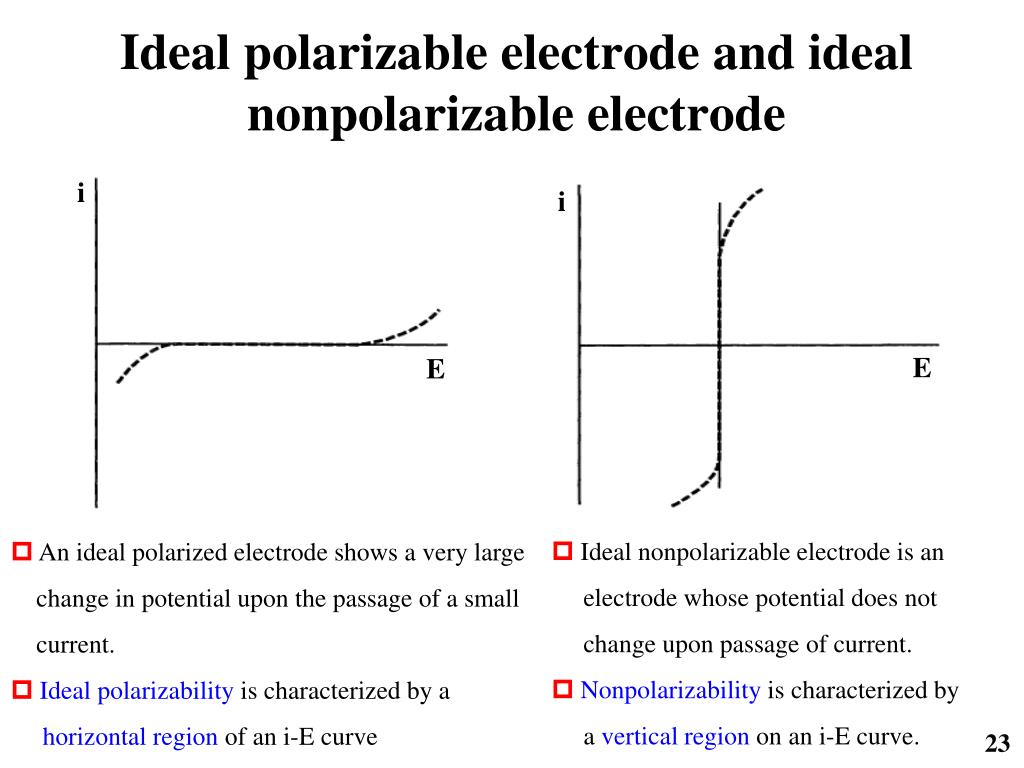
PPT - Electro-Optical Materials Laboratory Department of Chemical Engineering PowerPoint Presentation - ID:3931155

PDF) The surface phase at the ideal polarized mercury electrode2. Coulostatic measurements at the Hg electrode in dilute NaF aqueous solutions at various temperatures | Mathews Mulenga - Academia.edu
CHM 432: ADVANCED ELECTROCHEMISTRY CONTENTS 1.0. ELECTRICAL DOUBLE LAYER • Faradaic and Non faradaic current • Mass Transfe

SOLVED: 47 00/0 Question 9/18 (1 p ) Answer is mandatory In an electrochemical cell, if the potential of one of the electrodes was changing even when the current is kept constant (

SOLVED: In a electrochemical cell; if one ofthe electrodes was subjected to small currents, ad its potential was Ilound to stay constant then this electrode Is Indicator eleciode Ideall non-polarized elecirode Ideal
America's Smartest City by Movoto Blog Rated Most Livable City by Places Rated Almanac and The Economist Named among Best in the World Places to Visit. - ppt download