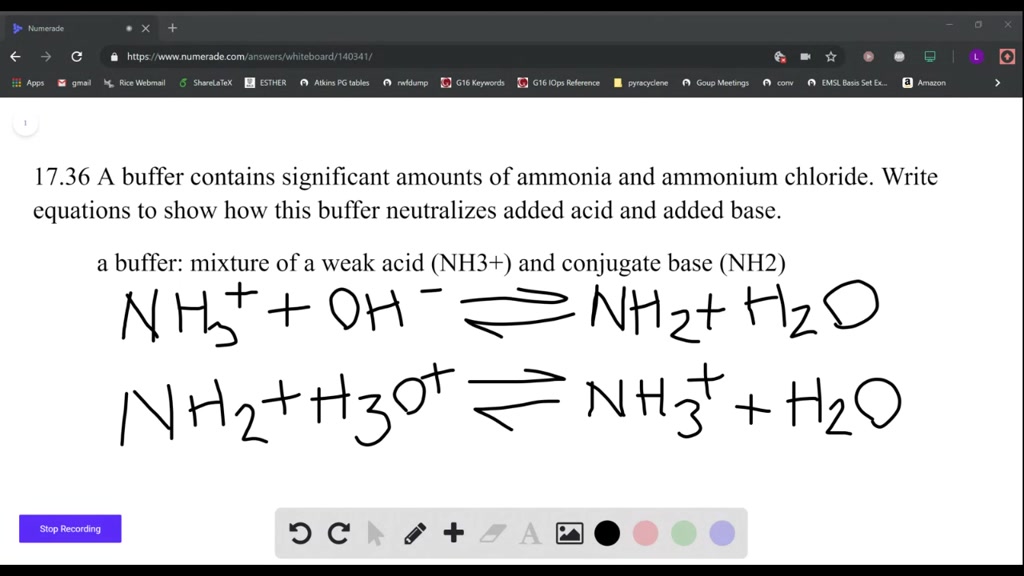
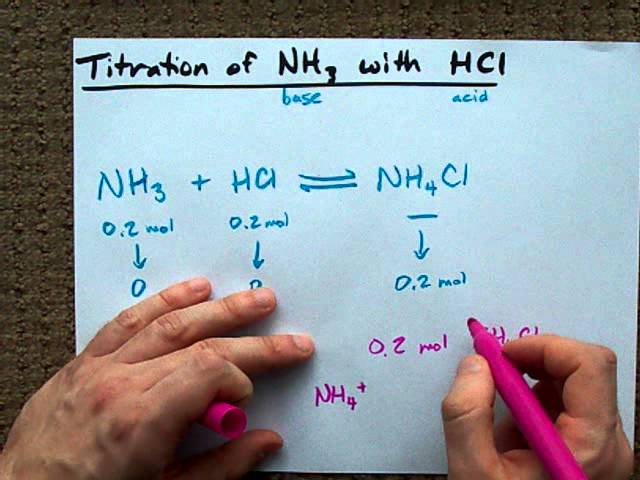
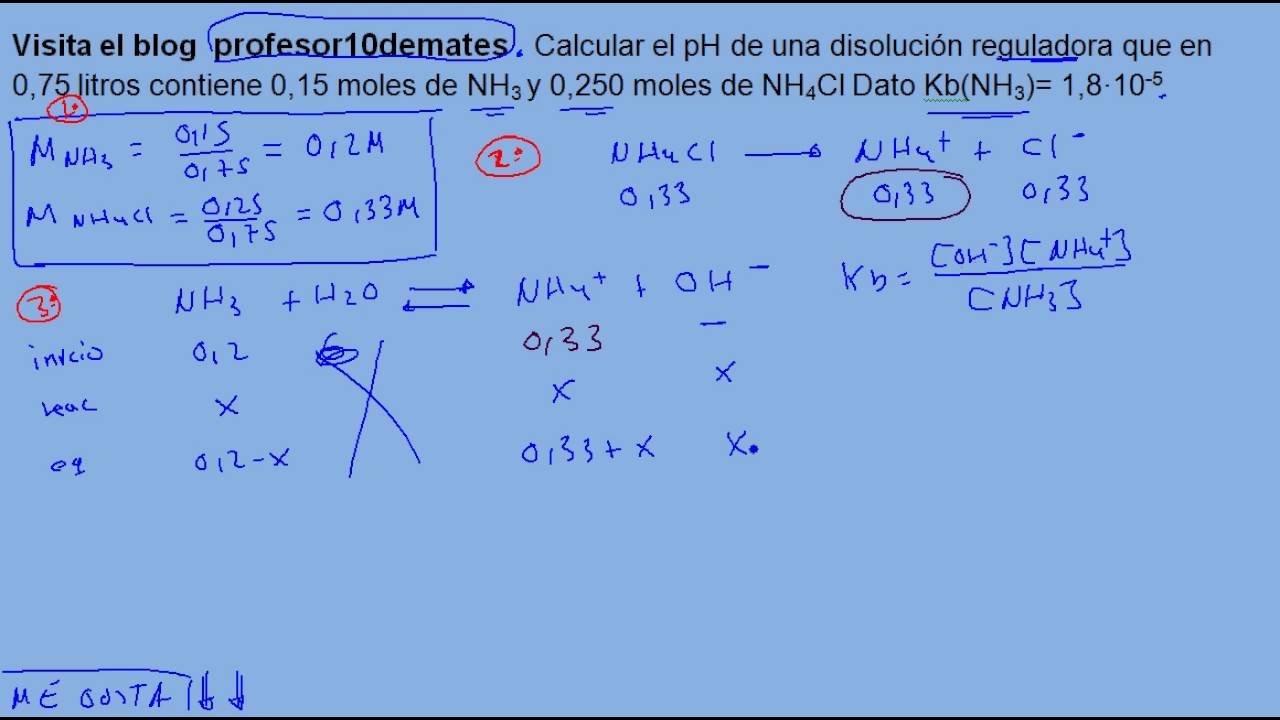
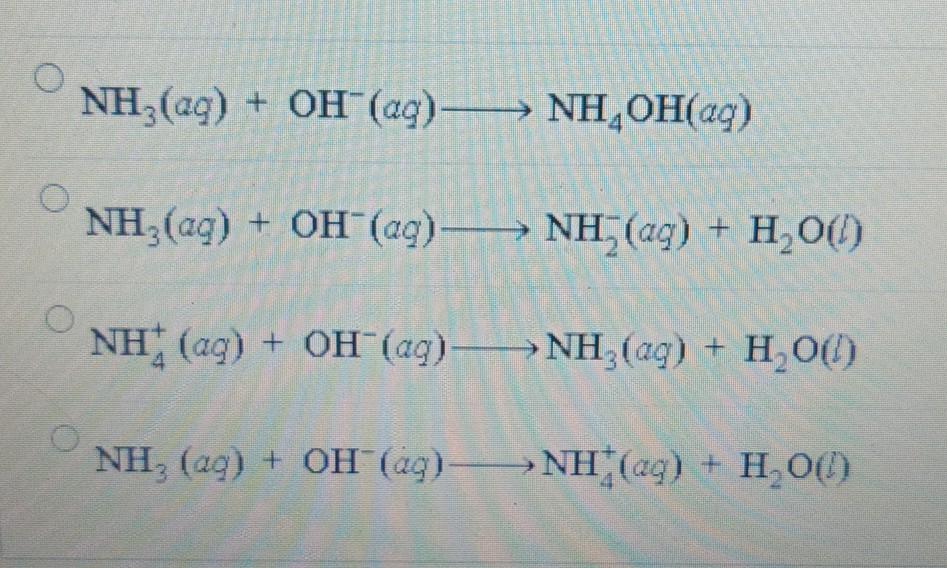
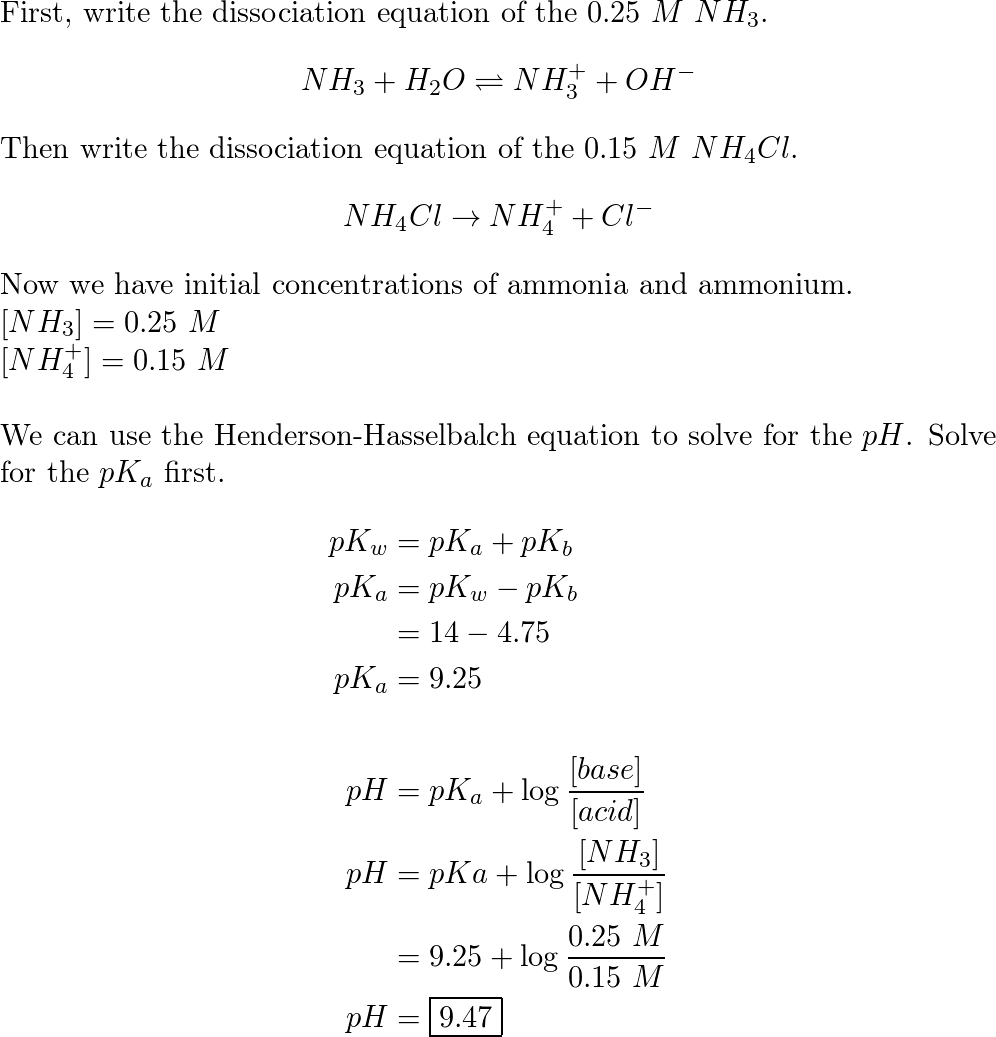OneClass: Calculate the pH of a .20M NH3/.20M NH4Cl buffer after the addition of 20.0mL of 0.10M HCl ...

A buffer solution containing NH3 and NH4Cl has a pH value of 9. pKb for NH3 is 4.7. If in the buffer solution total concentration of buffering reagents is 0.6 mol L^(-1),

PPT – Calculate the pH of the 0.30 M NH3/0.36 M NH4Cl buffer system. What is the pH after the addition of 20.0 mL of 0.050 M NaOH to 80.0 mL of
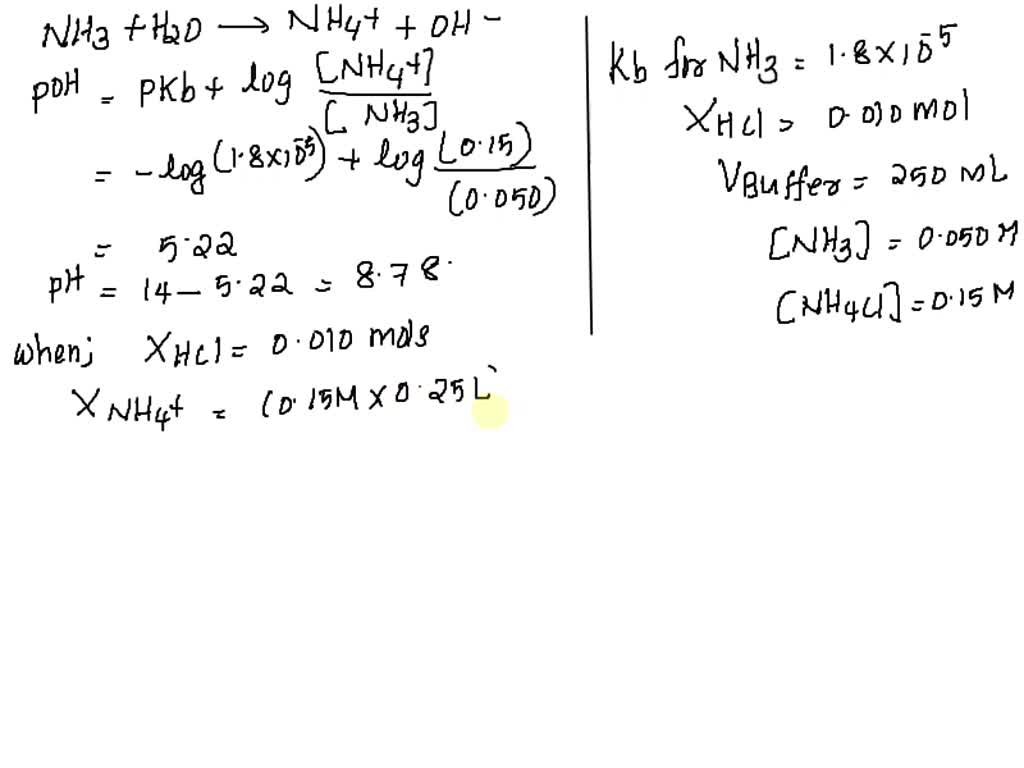
SOLVED: Calculate the pH after 0.010 mol HCl is added to 250.0 mL of a buffer solution that is 0.050 M NH3 and 0.15 M NH4Cl. (ammonia, Kb = 1.8×10−5)

a buffer solution contain NH3 and NH4CL total concentration of buffering agent is 0 6 molar if the pOH of - Chemistry - Equilibrium - 14195039 | Meritnation.com