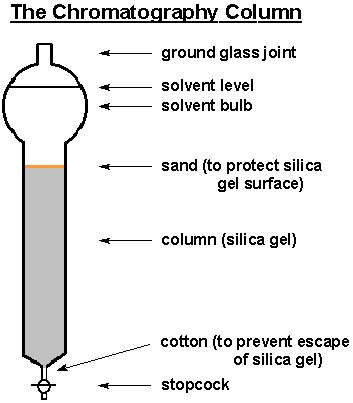
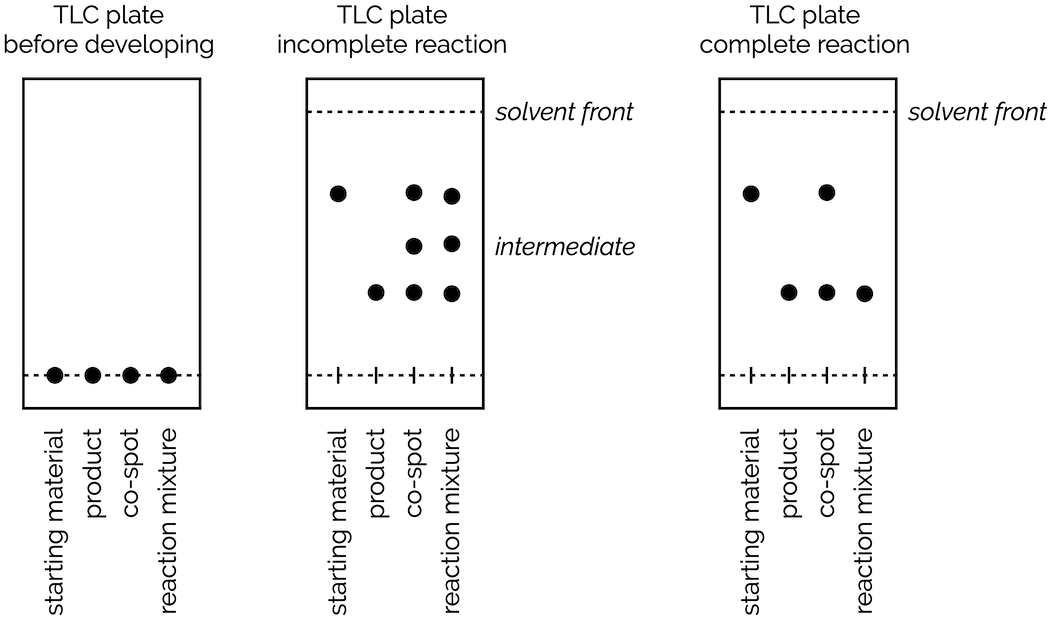
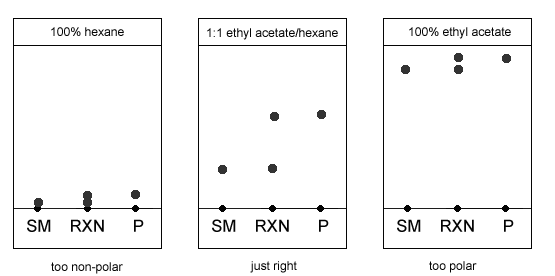
TLC and solvent polarity!! What does it mean that it's 'too non polar' and close to the origin? : r/Mcat

Do more polar molecules tend to have larger or smaller Rf values by TLC when compared to less polar molecules? | Homework.Study.com

Suppose you are trying to separate a mixture of three compounds with the help of TLC. Compound C is of medium polarity. Compounds A and B are of high polarity, but A
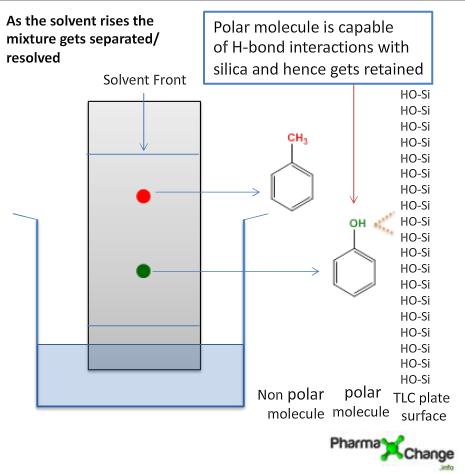
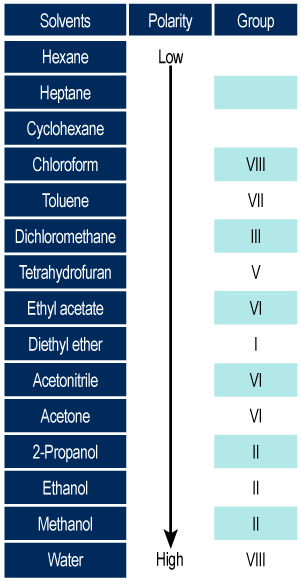



/chapter4/pages3and4/page3and4_files/rfvalue.png)
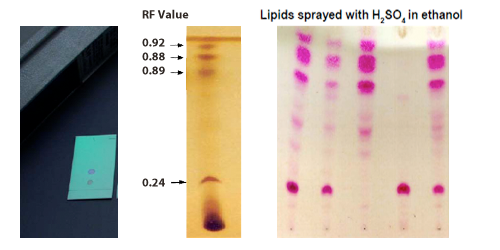




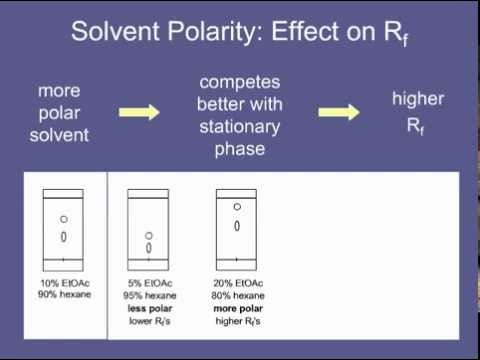


/chapter4/pages3and4/page3and4_files/tlcsetup.png)